GLYPHOSATE IN WATER AND WASTE WATER IN AFRICA
Interfacial phenomena Scientist.
AUTHOR: EDWIN OPONDO
MSc ENVIRONMENTAL ENGINEERING AND MANAGEMENT
JOMO KENYATTA UNIVERSITY OF AGRICULTURE AND TECHNOLOGY
JANUARY, 2026
SUBMITTED FOR PUBLICATION TO: DR. HANDA-CORRIGAN U.K BSc, MSc, PhD, Gulbenkian prize nominee 2025) FOUNDER environyoutheducation.co.uk
Cynthia Masanga – a BSc climate change student in Maseno university-Kenya

Abstract
Glyphosate (N-(phosphonomethyl)glycine) is the most widely applied herbicide globally and has been increasingly detected in surface and groundwater due to intensive agricultural and non-agricultural use. This manuscript reviews the biochemical properties and environmental behavior of glyphosate, pathways of entry into aquatic systems (including runoff, erosion, and urban discharge), its principal metabolite aminomethylphosphonic acid (AMPA), and the implications for water treatment and public health in African contexts. Evidence indicates strong adsorption to soil mineral phases, pH-dependent mobility, and microbial degradation to AMPA. However, limited infrastructure, seasonal rainfall patterns, and regulatory gaps in many African countries exacerbate the risk of glyphosate contamination. Recommendations include strengthened monitoring, upgraded wastewater treatment, harmonized regulation, and adoption of sustainable agricultural practices.
1. Introduction
Glyphosate (N-(phosphonomethyl)glycine) is a small, highly polar amino-phosphonate herbicide widely used for post-emergent weed control. Its physicochemical and biochemical characteristics dictate its environmental distribution, reactivity, and persistence (Borggaard & Gimsing, 2008; ATSDR, 2019). In Africa, rapid agricultural intensification, limited wastewater treatment infrastructure, and weak regulatory oversight have intensified concerns regarding glyphosate contamination of water resources.
2. Biochemical and Environmental Properties of Glyphosate
2.1 Acid–Base Speciation and Molecular Structure
Glyphosate contains a carboxyl group (-COOH), a phosphonic acid group (–PO3H2), and a secondary amine (-NH-), rendering it amphoteric. Four dissociation constants (reported approximately as pKa <2, 2.6, 5.6, and 10.6) govern its protonation states, leading to zwitterionic and anionic species across environmental pH ranges (Borggaard & Gimsing, 2008; Giesy, Dobson, & Solomon, 2000). This speciation affects solubility, sorption, and complexation behavior.
2.2 Hydrophilicity and Solubility
Glyphosate is highly water-soluble, with an octanol water partition coefficient (log Kow) typically reported between -3.2 and 2.8 and a solubility of approximately 10-12 g·L-1 for the free acid form (ATSDR, 2019). Such properties confer low potential for bioaccumulation and a preference for aqueous phases over lipid-rich media (Kanissery et al., 2019).
2.3 Metal Complexation and Adsorption to Soil Minerals
The phosphonate and carboxylate functionalities in glyphosate can form stable innersphere complexes with multivalent cations (e.g., Fe3+, Al3+, Ca2+, Mg2+, Mn2+), facilitating strong adsorption to iron and aluminum oxides as well as clay minerals (Gimsing et al., 2004; Barja & Dos Santos Afonso, 2005; Liu, Xu, & Sun, 2018). Adsorption occurs through ligand-exchange mechanisms, which often lead to relatively irreversible binding and decreased leaching potential, particularly in acidic, metal-rich soils (Prata et al., 2003).
2.4 Influence of pH, Competitive Anions, Ionic Strength, and Organic Matter
Glyphosate adsorption is strongly pH-dependent. Under acidic conditions, mineral surfaces are more positively charged, enhancing retention; as pH increases, glyphosate becomes more anionic and electrostatic repulsion reduces sorption (Gimsing & Borggaard, 2001). Competing anions such as phosphate and sulfate can displace glyphosate from adsorption sites via ligand exchange, while dissolved organic matter (DOM) may either inhibit adsorption by covering surface sites or promote mobility via complexation (Ololade et al., 2013; Borggaard & Gimsing, 2008).
2.5 Biodegradation and Environmental Fate
Microbial degradation is the principal dissipation route for glyphosate, primarily yielding aminomethylphosphonic acid (AMPA) following C–P bond cleavage (Forlani et al., 1999; Sviridov et al., 2015). Reported aerobic soil half-lives vary from days to several weeks, influenced by microbial community composition, temperature, moisture, and organic carbon content (Giesy et al., 2000). Adsorption to mineral phases can limit 6 microbial accessibility and slow degradation, while in aquatic systems glyphosate frequently partitions to suspended particulates and sediments.
2.6 Biochemical Mode of Action
Glyphosate inhibits 5-enolpyruvyl-shikimate-3-phosphate synthase (EPSPS), a key enzyme in the shikimate pathway for biosynthesis of aromatic amino acids in plants and many microorganisms (Steinrücken & Amrhein, 1980). By occupying the phosphoenolpyruvate (PEP) binding site, glyphosate acts as a competitive inhibitor, preventing formation of 5-enolpyruvyl-shikimate-3-phosphate (EPSP) and arresting downstream biosynthetic processes (Duke & Powles, 2008).
Glyphosate N-(phosphonomethyl)glycine (C₃H₈NO₅P) Glyphosate is basically glycine with a phosphonomethyl group attached to the nitrogen.
Aminomethylphosphonic acid (AMPA) CH₆NO₃P
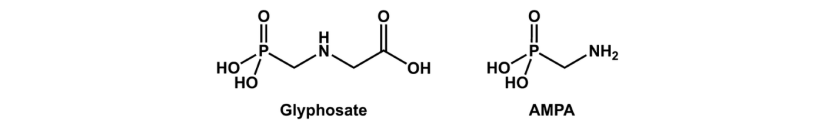
Relationship between the two Glyphosate degrades into AMPA
Glyphosate (C₃H₈NO₅P)+H₂O→AMPA (CH₆NO₃P)+Glycine (C₂H₅NO₂)
Characteristics Of Glyphosate And AMPA:
Glyphosate/aminomethylphosphonic acid and human health
Glyphosate, a common herbicide, and its metabolite AMPA are frequently detected in human urine and the environment as exposure markers (Chaiklieng & Uengchuen, 2020). Early reviews found low absorption, minimal bioaccumulation, and no clear human carcinogenic or reproductive risk at typical exposure levels (Williams, Kroes & Munro, 2000), though the 2000 safety study was retracted in 2025, raising new concerns. Some epidemiological studies link glyphosate/AMPA to liver inflammation, metabolic disturbances, and possibly higher breast cancer risk, but causal links remain unclear (CHAMACOS cohort, 2021; Franke et al., 2022). Toxicological studies show high doses can cause DNA damage or cytotoxicity in vitro, yet typical exposures are generally low-risk (Koller et al., 2012; EFSA, 2015). Overall, scientific opinion remains divided on long-term, low-dose effects.
3. Pathways of Entry into Aquatic Systems
Glyphosate enters aquatic systems via diffuse (non-point) sources such as runoff and erosion following application, and via point sources including equipment wash-off and urban discharge (Botta et al., 2009; Hanke et al., 2010). In urban settings, application to impervious surfaces increases direct runoff into stormwater systems, reducing soilmediated attenuation and facilitating transport into receiving waters.
4. Environmental Evidence and Monitoring in Africa
Although monitoring in Africa remains sparse, available studies report detection of glyphosate and AMPA in surface waters and sediments in several countries, commonly associated with agricultural runoff and wastewater inputs (Horn et al., 2019; Botta et al., 2009). Detection frequencies and concentrations are influenced by sampling timing 8 relative to application and regional hydrology. The persistence of AMPA in sediments raises concerns regarding long-term ecological impacts.
5. Human Health and Ecological Risks
Human exposure to glyphosate occurs through ingestion of contaminated water and food, inhalation of spray drift, and dermal contact. Epidemiological and toxicological studies have raised concerns about potential carcinogenicity, genotoxicity, and organ-specific toxicity, culminating in the International Agency for Research on Cancer (IARC) classification of glyphosate as 'probably carcinogenic to humans' (IARC, 2015). Other reviews emphasize low vertebrate toxicity but note broader ecological risks and sublethal effects on non-target organisms (Myers et al., 2016; Giesy et al., 2000).
6. Regulatory Framework and Gaps
Regulatory standards for glyphosate in water vary internationally. The European Union enforces a 0.1 µg·L-1 limit for individual pesticides in drinking water, whereas other jurisdictions set higher values for glyphosate specifically (e.g., EPA tolerances for residues). Many African countries lack harmonized standards and adequate laboratory capacity for routine glyphosate monitoring, creating regulatory gaps that hinder effective management and public health protection (Hanke et al., 2010).
7. Mitigation Strategies and Recommendations
To mitigate glyphosate contamination, an integrated approach is required:
8. Conclusion
Glyphosate’s chemical properties high water solubility, amphoteric speciation, and strong metal complexation dictate its environmental behavior. In African contexts, insufficient treatment infrastructure, variable land management practices, and regulatory gaps elevate the risk of water contamination. Strengthened monitoring, targeted mitigation, and policy reform are necessary to safeguard water security and ecosystem integrity.
References
